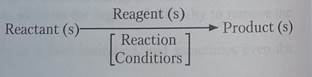Chemical properties of elements
Vanderwaals radius
Even when two atoms that are near one another will not bind, they will still attract one another. This phenomenon is known as the Vanderwaals interaction. The Vanderwaals forces cause a force between the two atoms. This force becomes stronger, as the atoms come closer together. However, when the two atoms draw too near each other a rejecting force will take action, as a consequence of the exceeding rejection between the negatively charged electrons of both atoms. As a result, a certain distance will develop between the two atoms, which is commonly known as the Vanderwaals radius. Through comparison of Vanderwaals radiuses of several different pairs of atoms, we have developed a system of Vanderwaals radiuses, through which we can predict the Vanderwaals radius between two atoms, through addition.
Ionic radius
The radius that an ion has in an ionic crystal, where the ions are packed together to a point where their outermost electronic orbitals are in contact with each other. An orbital is the area around an atom where, according to orbital theory, the probability of finding an electron is the greatest.
Isotopes
The atomic number does not determine the number of neutrons in an atomic core. As a result, the number of neutrons within an atom can vary. As a result, atoms that have the same atomic number may differ in atomic mass. Atoms of the same element that differ in atomic mass are called isotopes. Mainly with the heavier atoms that have a higher atomic number, the number of neutrons within the core may exceed the number of protons. Isotopes of the same element are often found in nature alternately or in mixtures.
An example: chlorine has an atomic number of 17, which basically means that all chlorine atoms contain 17 protons within their core. There are two isotopes. Three-quarters of the chlorine atoms found in nature contain 18 neutrons and one quarter contains 20 neutrons. The mass numbers of these isotopes are 17 + 18 = 35 and 17 + 20 = 37. The isotopes are written as follows: 35C1 and 37C1. When isotopes are noted this way the number of protons and neutrons does not have to be mentioned separately, because the symbol of chlorine within the periodic chart (CI) is set on the seventeenth place. This already indicates the number of protons, so that one can always calculate the number of neutrons easily by means of the mass number.
A great number of isotopes is not stable. They will fall apart during radioactive decay processes. Isotopes that are radioactive are called radioisotopes.
Electronic shell
The electronic configuration of an atom is a description of the arrangement of electrons in circles around the core. These circles are not exactly round; they contain a wave-like pattern. For each circle the probability of an electron to be present on a certain location is described by a mathematic formula. Each one of the circles has a certain level of energy, compared to the core. Commonly the energy levels of electrons are higher when they are further away from the core, but because of their charges, electrons can also influence each another's energy levels. Usually the middle circles are filled up first, but there may be exceptions due to rejections. The circles are divided up in shells and sub shells, which can be numbered by means of quantities.
Energy of first ionization
The ionization energy means the energy that is required to make a free atom or molecule lose an electron in a vacuum. In other words; the energy of ionisation is a measure for the strength of electron bonds to molecules. This concerns only the electrons in the outer circule.
Besides the energy of the first ionization, which indicates how difficult it is to remove the first electron from an atom, there is also an energy measure for second ionization. This energy of second ionization indicates the degree of difficulty to remove the second atom.
As such, there is also the energy of a third ionization, and sometimes even the energy of a fourth or fifth ionization.
IV. Translate the following text into English using proper terms given below:
Что такое сырая нефть? Нефть - это не химический элемент, а смесь соединений.
Интересные свойства нефти проявляются при нагревании. Если нагреть нефть до температуры кипения и выдержать ее в этом состоянии некоторое время, то она испарится, но не полностью.
Для сравнения возьмем воду. Нагреем сосуд с водой до 1000 С. Если продолжить нагрев, то вода начнет испаряться и через некоторое время выкипит полностью. И температура при этом сохранится на прежнем уровне.
Чтобы всё стало понятно, возьмём тот же сосуд и заполним его сырой нефтью средней плотности. Начнем нагревать нефть. Когда температура достигнет 65° С, сырая нефть закипит. Мы продолжаем нагревание, но при этом поддерживаем температуру на том же уровне. Через некоторое время нефть перестанет кипеть.
Следующий шаг - поднять температуру до 230°С. Нефть начнет испаряться вновь, но спустя несколько минут испарение прекратится. Процесс нужно продолжать, параллельно повышая температуру нагрева (т.е. до 65,230,400 и 480°С).
Это подтверждает сложный состав сырой нефти, которая состоит из определённых сочетаний атомов углерода и водорода, и которые называются углеводородами. Каждое из этих соединений характеризуется своей собственной температурой кипения.
Вот на этом основывается процесс переработки сырой нефти, называемый нефтепегонкой. Но при выходе из нефтяной скважины нефть содержит частицы горных пород, воду, а также растворенные в ней соли и газы. Эти примеси вызывают коррозию оборудования и серьезные затруднения при транспортировке и переработке нефтяного сырья. Таким образом, для экспорта или доставки в отдаленные от мест добычи нефтеперерабатывающие заводы необходима ее промышленная обработка: из нее удаляется вода, механические примеси, соли и твердые углеводороды, выделяется газ.
Terms and word combinations:
Impurity, processing, vessel, rock particles, crude oil, oil well, chemical compound, refinery plant, oil distillation, oil extraction, solid hydrocarbons, compound mixture,boiling temperature
LESSON #3
CHEMICAL REACTIVITY
Organic chemistry encompasses a very large number of compounds (many millions).
We begin by defining some basic terms that will be used frequently as this subject is elaborated.
Chemical Reaction: A transformation resulting in a change of composition, constitution and/or configuration of a compound (referred to as the reactant or substrate).
Reactant or Substrate: The organic compound undergoing change in a chemical reaction. Other compounds may also be involved, and common reactive partners (reagents) may be identified. The reactant is often (but not always) the larger and more complex molecule in the reacting system. Most (or all) of the reactant molecule is normally incorporated as part of the product molecule.
Reagent:A common partner of the reactant in many chemical reactions. It may be organic or inorganic; small or large; gas, liquid or solid. The portion of a reagent that ends up being incorporated in the product may range from all to very little or none
Product (s): The final form taken by the major reactant(s) of a reaction.
Reaction Conditions:The environmental conditions, such as temperature, pressure, catalysts & solvent, under which a reaction progresses optimally. Catalysts are substances that accelerate the rate (velocity) of a chemical reaction without themselves being consumed or appearing as part of the reaction product. Catalysts do not change equilibria positions.
Chemical reactions are commonly
written as equations: