| ADDITION |
| 4 + 7 = 11 | · 4 plus 7 equals 11 · 4 plus 4 is equal to 11 · 4 added to 7 makes 11 · 4 and 7 is 11 · 4 and 7 are 11 |
| SUBTRACTION |
| 11 – 4 = 7 | · 11 minus 4 equals 7 · 4 from 11 leaves 7 · 11 diminished by 4 is equal to 7 |
| MULTIPLICATION |
| 1 × 1 = 1 2 × 2 = 4 3 × 3 = 9 4 × 4 = 16 | · Once one is one. · Twice two is four. · Three times three is nine. · Four times four is sixteen. |
| 12 × 12 = 144 | 12 (multiplied) by 12 equals 144 |
| DIVISION |
| 25 : 5 = 5 | 25 divided by 5 equals 5 |
| DECIMAL FRACTIONS |
| 0.2 | · Point two · Nought point two · Zero point two · Point two |
| 0.002 | · 0 point 00 two · Point two oes two · Point nought nought two |
| 1.1 | one point one |
| 1.25 | one point two five |
| 63.57 | sixty-three point five seven |
| INVOLUTION |
| 32 | · Three squared (Three square) · Three (raised) to the second power · Three to the power two · The second power of three |
| 53 | · Five cubed · Five cube · Five (raised) to the third power · Five to the power three · The third power of five |
| 107 | Ten to the seventh power |
| 10-7 | Ten to the minus seventh power |
| RATIO |
| 1 : 2 | The ratio of one to two |
| 4 : 2 = 2 | The ration of four to two is two |
| 20 : 5 = 16 : 4 | · The ratio of twenty to five equals the ratio of sixteen to four · Twenty is to five as sixteen is to four |
| CHEMICAL ELEMENTS |
| No. | Symbol | Latin name | English name |
| | H | hydrogenium | hydrogen |
| | He | helium | helium |
| | Li | litium | lithium |
| | Be | beryllium | beryllium |
| | B | borum | boron |
| | C | carboneum | carbon |
| | N | nitrogenium | nitrogen |
| | O | oxygenium | oxygen |
| | F | fluorum | fluorine |
| | Ne | neon | neon |
| | Na | natrium | sodium |
| | Mg | magnesium | magnesium |
| | Al | aluminium | aluminum aluminium |
| | Si | silicium | silicon |
| | P | phosphorus | phosphorus |
| | S | sulphur | sulphur |
| | Cl | chlorum | chlorine |
| | Ar | argon | argon |
| | K | kalium | potassium |
| | Ca | calcium | calcium |
| | Sc | scandium | scandium |
| | Ti | titanium | titanium |
| | V | vanadium | vanadium |
| | Cr | chromium | chromium |
| | Mn | manganum | manganese |
| | Fe | ferrum | iron |
| | Co | cobaltum | cobalt |
| | Ni | niccolum | nickel |
| | Cu | cuprum | copper |
| | Zn | zincum | zinc |
| | Ga | gallium | gallium |
| | Ge | germanium | germanium |
| | As | arsenicum | arsenic |
| | Se | selenium | selenium |
| | Br | bromum | bromine |
| | Kr | krypton | krypton |
| | Rb | rubidium | rubidium |
| | Sr | strontium | strontium |
| | Y | yttrium | yttrium |
| | Zr | zirconium | zirconium |
| | Nb | niobium | niobium |
| | Mo | molybdaenum | molybdenum |
| | Tc | technetium | technetium |
| | Ru | ruthenium | ruthenium |
| | Rb | rhodium | rhodium |
| | Pd | palladium | palladium |
| | Ag | argentum | silver |
| | Cd | cadmium | cadmium |
| | In | indium | indium |
| | Sn | stannum | tin |
| | Sb | stibium | antimony |
| | Te | tellurium | tellurium |
| | I | iodium | iodine |
| | Xe | xenon | xenon |
| | Cs | caesium | cesium caesium |
| | Ba | barium | barium |
| | La | lanthanum | lanthanum |
| | Ce | cerium | cerium |
| | Pr | praseodymium | praseodymium |
| | Nd | neodymium | neodymium |
| | Pm | promethium | promethium |
| | Sm | samarium | samarium |
| | Eu | europium | europium |
| | Gd | gadolinium | gadolinium |
| | Tb | terbium | terbium |
| | Dy | dysprosium | dysprosium |
| | Ho | holmium | holmium |
| | Er | erbium | erbium |
| | Tm | thulium | thulium |
| | Yb | ytterbium | ytterbium |
| | Lu | lutetium | lutetium |
| | Hf | hafnium | hafnium |
| | Ta | tantalum | tantalum |
| | W | wolframium | tungsten wolfram |
| | Re | rhenium | rhenium |
| | Os | osmium | osmium |
| | Ir | iridium | iridium |
| | Pt | platinum | platinum |
| | Au | aurum | gold |
| | Hg | hydrargyrum | mercury |
| | Tl | thallium | thallium |
| | Pb | plumbum | lead |
| | Bi | bismuthum | bismuth |
| | Po | polonium | polonium |
| | At | astatium | astatine |
| | Rn | radon | radon |
| | Fr | francium | francium |
| | Ra | radium | radium |
| | Ac | actinium | actinium |
| | Th | thorium | thorium |
| | Pa | protactinium | protactinium |
| | U | uranium | uranium |
| | Np | neptunium | neptunium |
| | Pu | plutonium | plutonium |
| | Am | americium | americium |
| | Cm | curium | curium |
| Chemical compounds and Chemical equations |
| 2MnO2 | two molecules of M N O two |
| H+ | · hydrogen ion · univalent positive hydrogen ion |
| Cu++ | divalent positive cuprum ion |
| Al+++ | trivalent positive aluminium ion |
| Cl- | · negative chlorine ion · negative univalent chlorine ion |
| O = C = O | C O two |
| 4KCl | four molecules of K C L |
| 4HCl + O2 = 2Cl2 + 2H2O | four molecules of H C L plus O two give two molecules of C L two and two molecules of H two O |
| Zn + CuSO4 = Cu + ZnSO4 | Z N plus C U S O four give C U plus Z N S O four |
| PCl3 + 2Cl → PCl5 | P C L three plus two molecules of C L give P C L five |
H2 + J2 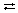 2HJ 2HJ | H two plus J two form and are formed from two molecules of H J |
| C2H2 + H2O → CH3CHO | C two H two plus H two O give C H three C H O |
AcOH 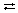 AcO- + H+ AcO- + H+ | A C O H forms and is formed from A C oxygen ion plus hydrogen ion |
| | | | | |
Наши рекомендации
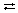 2HJ
2HJ 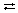 AcO- + H+
AcO- + H+ 