Qualitative and quantitative analysis of the combustion products.
When burned with excess air a  1produkty combustion are composed of carbon dioxide CO2. H2O, water vapor, nitrogen N2 and oxygen O2 free.
1produkty combustion are composed of carbon dioxide CO2. H2O, water vapor, nitrogen N2 and oxygen O2 free.
Quantitatively, the combustion of 1 kg of fuel, will have products of combustion (in moles):
Carbon dioxide . . …. М со = 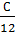 mole/kg
mole/kg
Steam water . . . … М н о =  mole/kg
mole/kg
Nitrogen …. . . МN = 0,79а Lo mole/kg
Oxygen . . . . . Мо = 0,21(а-1) Lo mole/kg
Total combustion: М2 = 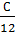 +
+ 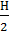 + 0,79а Lo + 0,21(а-1) Lo
+ 0,79а Lo + 0,21(а-1) Lo
Transforming the result obtained by adding and subtracting 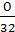 mole:
mole:
М2 = 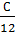 +
+ 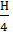 -
- 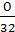 +
+ 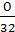 +
+ 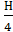 0,79а Lo + 0,21(а-1) Lo mole/kg
0,79а Lo + 0,21(а-1) Lo mole/kg
The first three terms give ( 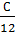 +
+ 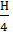 -
- 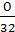 ) gives the number of moles of oxygen theoretically required for combustion of 1 kg of fuel.
) gives the number of moles of oxygen theoretically required for combustion of 1 kg of fuel.
( 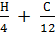 -
- 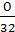 ) = 0,21 Lo
) = 0,21 Lo
Consequently moles М2 = а Lo + 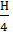 +
+ 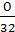 mole/kg
mole/kg
At а 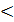 1 in the flue gas will be stored CO2 CO, H2O, N2; free oxygen is not. To determine the number of products of combustion at a <1 is necessary to determine how much (in kilograms) of carbon monoxide in fuel burn. Equation connection between the amount of oxygen that has gone on a chemical reaction in the air we have.
1 in the flue gas will be stored CO2 CO, H2O, N2; free oxygen is not. To determine the number of products of combustion at a <1 is necessary to determine how much (in kilograms) of carbon monoxide in fuel burn. Equation connection between the amount of oxygen that has gone on a chemical reaction in the air we have.
а ( 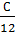 +
+ 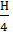 -
- 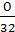 ) mole oxygen
) mole oxygen
in the fuel available 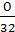 mole oxygen. The total amount of oxygen is equal to
mole oxygen. The total amount of oxygen is equal to
а ( 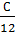 +
+ 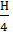 -
- 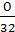 )+
)+ 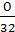 mole.
mole.
Assume that C kg of carbon containing 1 kg of fuel. With burns to CO2 and C with carbon burn in kg CO: С = С со + С
Then, on the combustion reaction will be spent
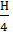 +
+ 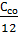 +
+ 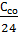 - mole oxygen.
- mole oxygen.
Therefore а ( 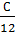 +
+ 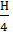 -
- 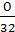 )+
)+ 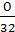 =
= 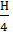 +
+ 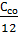 +
+ 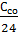
Replacing C with through (C – Cco), we have
а ( 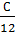 +
+ 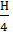 -
- 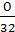 )+
)+ 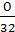 =
= 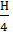 +
+ 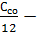
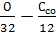 +
+ 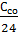
as ( 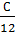 +
+ 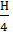 -
- 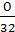 ) = 0,21 Lo
) = 0,21 Lo
the 0,21а Lo = 0,21а Lo - 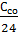
from whence С со = 24*0,21(1-а) Lo = 5,04 (1-а) Lo kg/kg (26)
Knowing the relationship between a and the amount of carbon with C, combusted into carbon dioxide, it is possible to determine the amount of combustion products at а 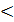 1:
1:
Steam water М НО =  mole/kg
mole/kg
carbon dioxide М С 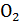 =
= 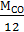 =
= 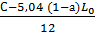 mole/kg
mole/kg
carbon monoxide М СО = 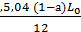 mole/kg
mole/kg
nitrogen МN = 0,79а Lo mole/kg
Total combustion:
М2 =  +
+ 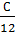
Transforming this expression. Adding and subtracting from the right side 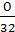 + 0,21а Lo we get:
+ 0,21а Lo we get:
М2 =  +
+ 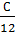 -
- 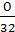 +
+ 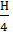 +
+ 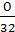 + 0,79а Lo + 0,21а Lo - 0,21а Lo :
+ 0,79а Lo + 0,21а Lo - 0,21а Lo :
as
( 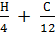 -
- 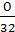 ) = 0,21 Lo mole/kg
) = 0,21 Lo mole/kg
the
М2 = 0,79а Lo + 0,21а Lo - 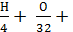 0,21а Lo - 0,21а Lo
0,21а Lo - 0,21а Lo
or
М2 = а Lo + 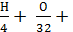 0,21(1-а) Lo (28)
0,21(1-а) Lo (28)
As can be seen from the expressions (25) and (28), the number of moles M2 of combustion products different from the number of moles of Lo and fresh charge air.
Changing the number of moles of gases in the combustion process is characterized by the ratio of the molecular changes. Chemical changes in the molecular ratio m0 called the ratio of the number of moles of the combustion products to the number of moles of the fresh charge. m0 = 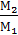 .
.
When the engine is in its cylinder is always a certain amount of M r of the residual gases. (M2 + Mr) is called the real coefficient of the molecular changes m.
m = 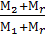
Week of the numerator and denominator in this expression on the M1, we get:
m = 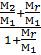 ,
,
ort
m = 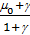 , (30)
, (30)
Where 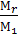 is the coefficient of residual gases g.
is the coefficient of residual gases g.
When the engine is running on rich mixture that is at а 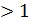 , work efficiency is reduced. For complete combustion of two moles of carbon requires two moles of oxygen. The reaction proceeds according to the formula
, work efficiency is reduced. For complete combustion of two moles of carbon requires two moles of oxygen. The reaction proceeds according to the formula
2С +2О2 = 2СО2 +188 800 kcal.
This combustion is allocated 188 800 kcal. If one is not enough oxygen moth, the combustion takes place according to the formula
2С + О2 = 2СО +52 000 kcal.
Thus, one is not enough oxygen moles, that leads to a loss of 136,000 kcal.
For 1 kg of combustion fuel required 0,21 Lo moles of oxygen; for a <1 a shortage of oxygen in moles will be:
МО2 =0,21Lo - 0,21а Lo =0,21(1-а) Lo mole,
heat loss DQ per 1 kg of fuel is:
DQ =136 800*0,21(1-а) Lo kcal /kg,
DQ =28700 (1-а) Lo kcal /kg, (31)
The calorific value (calorie) 1kg of gasoline, kerosene, diesel fuel, approximately equal to 10000 kcal / kg. The theoretical amount of air required Lo ≅ 0,5 moles / kg. For a = 0.9 heat loss equals.
DQ =28700 *0,1*0,5 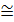 1435 kca /kg,
1435 kca /kg,
which is 14% fuel heat capacity.
Lecture 7
PHASE OF BURNING.
The internal combustion engine fuel combustion is not instantaneous; on the indicator diagram, shot in the coordinates "pressure p" - "angle of rotation of the shaft a», easy to install individual phases of combustion.
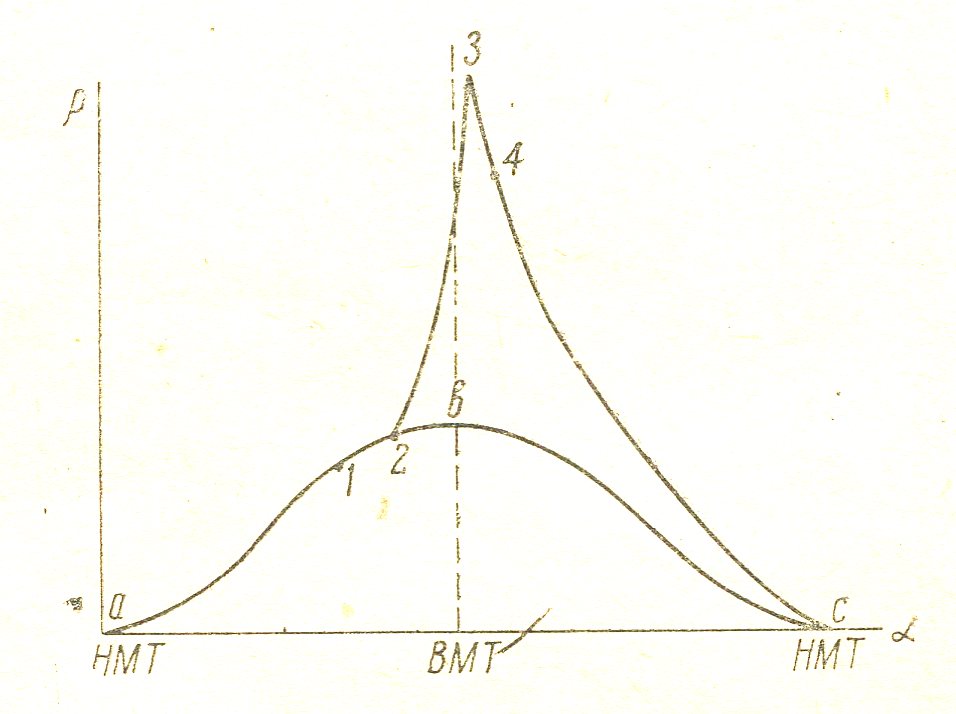
Figure 7
Figure 7 given indicator diagram for gasoline or gas engine. The curve abc shows the compression ab and expansion process bc when the ignition is off. Point 1 corresponds to the moment of ignition. As can be seen from the diagram, from the time of ignition until the combustion pressure rises and passes a certain interval 1-2, which is called the induction period. Induction period is the 1st phase of combustion. Line 2-3 shows the development of combustion, during which time the flame front propagates at a speed of about 20 m / s, covers the entire volume of the compressed combustible mixture. This second phase of combustion is accompanied by a rapid increase of pressure. The second phase is called the "visible burning." In fact, to achieve maximum combustion pressure of time does not stop and continues to expand the line to the point 4. Period 3-4 is burning phase, called "burn-phase"
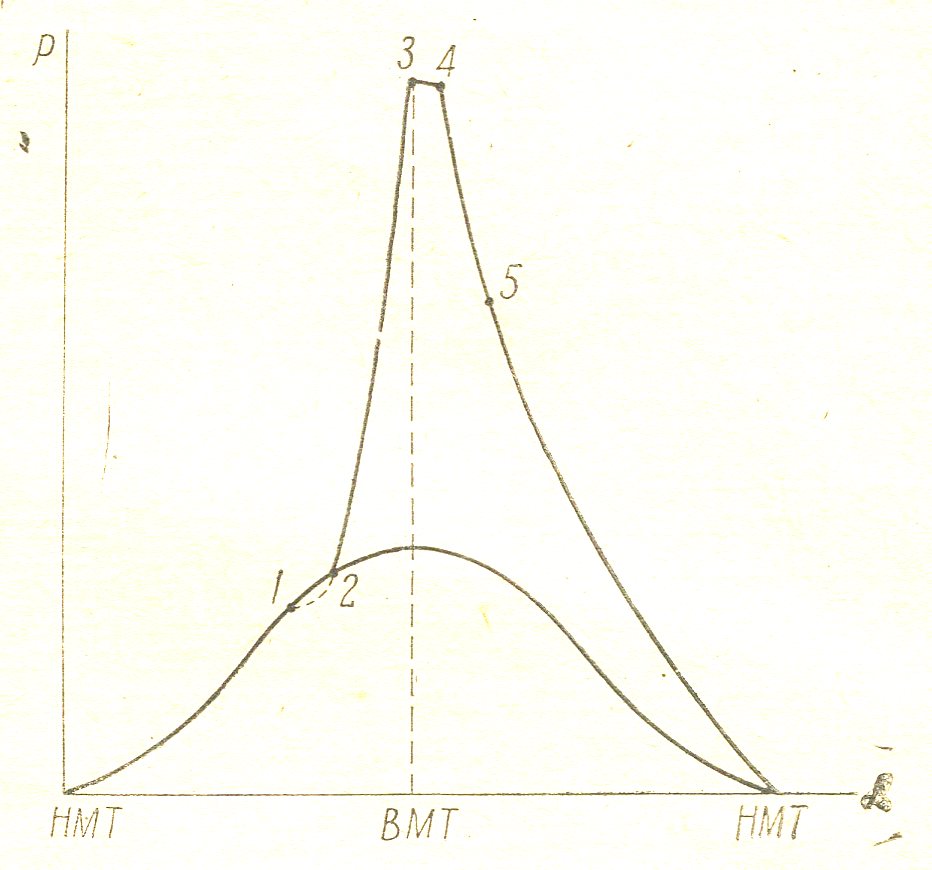
Figure 8
By extension line burns that part of the fuel that has not had time to burn down to the point 3. In addition, the burning of dissociation products. The molecules of the combustion products CO2 and H2O under the influence of high temperature, which by the end of combustion in gasoline engines reaches 27,000 abs and high pressure, are cleaved to form CO molecules, heat. With the expansion of gases when their temperature begins to drop, the process of combustion of the dissociation products. As is known, in gasoline engines combustion process takes place in three specific phases. The high-speed diesel engines burning process has four phases. FIG. 8 given indicator diagram of such an engine.
Point 1 corresponds to the beginning of injection; It passes a period of 1-2 before start burning. During this period, called the induction period, gas pressure is relatively more reduced due to the absorption of heat by heating the injected fuel drops. Point 2 corresponds to the start of visible combustion are any drops to the self-ignition temperature, ignited and lit the fuel that the pump has time to apply for the period of the first phase - induction. The flame front portions extend at approximately the same speed as the engine and the carburettor, and the combustion process through line 2-3 does not differ essentially from combustion in a carburettor engine. During the induction period, the pump has time to file into the cylinder a significant proportion of the total quantity of fuel. During the combustion of the fuel of the large heat release occurs, accompanied by an increase in pressure. 2-3 The second phase called "rapid combustion phase."
In point 3, the fuel supply is not the end, the pump continues to deliver fuel, but now do not need to drop heating to self-ignition temperature as the fuel droplets are injected not Wednesday compressed hot air, and on Wednesday the raging flame and light, the fuel can not accumulate in the cylinder. This part of the fuel combustion takes place without any appreciable change in pressure.
At the same time, the piston a few time to get away from TDC, which also entails a pressure drop. Therefore, lines 3-4 combustion occurs without a significant change in pressure, which can increase or more, or more drop. The third phase of 3-4 is called "combustion at constant pressure phase". In point 4, does not end with the burning of the fuel and burns for line extensions. Afterburning in diesels is much higher than in a carburetor engine afterburner of the fact that there are not only a mixture of fuel vapors and droplets. Afterburning ends at point 5.
At the same time, the piston a few time to get away from TDC, which also entails a pressure drop. Therefore, lines 3-4 combustion occurs without a significant change in pressure, which can increase or more, or more drop. The third phase of 3-4 is called "combustion at constant pressure phase". In point 4, does not end with the burning of the fuel and burns for line extensions.
Afterburning in diesels is much higher than in a carburetor engine afterburner of the fact that there are not only a mixture of fuel vapors and droplets. Afterburning ends at point 5.
4-5 The fourth phase is called "burnout phase." By expanding illuminated line and also the dissociation products in diesel engines but dissociation relatively less intense, since the temperature at point 4, reaching values on the order 23000S abs significantly below the temperature at the end of the combustion engine carburettor. Combustion process in diesel engines goes through four phases.
Lecture 6
