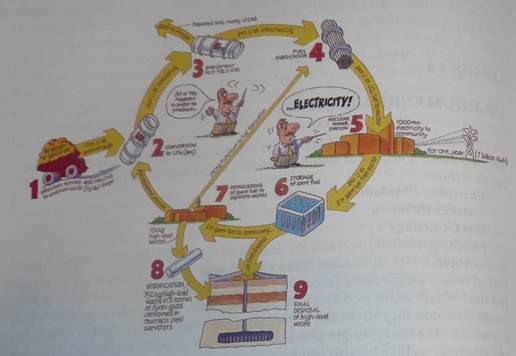
From uranium ore to reactor fuel
Uranium ore can be mined by underground or open-cut methods, depending on its depth. After mining, the ore is crushed and ground up. Then it is treated with acid to dissolve the uranium, which is then recovered from solution. Uranium may also be mined by in situ leaching (ISL), where it is dissolved from the orebody in situ and pumped to the surface. The end product of the mining and milling stages, or of ISL, is uranium oxide concentrate (U3 08). This is the form in which uranium is sold. Before it can be used in a reactor for electricity generation, however, it must undergo a series of processes to produce a useable fuel.
For most of the world's reactors, the next step in making useable fuel is to convert the uranium oxide into gas, uranium hexafluoride (UF^, which enables it to be enriched. Enrichment increases the proportion of the uranium-235 isotope from its natural level of 0.7% to 3 - 4%. This enables greater technical efficiency in reactor design and operation, particularly in larger reactors, and allows the use of ordinary water as a moderator. After enrichment, the UF6 gas is converted to uranium dioxide (U02) which is formed into fuel pellets. These fuel pellets are placed inside thin metal tubes which are assembled in bundles to become the fuel elements for the core of the reactor. For reactors which use natural uranium as their fuel (and hence which require graphite or heavy water as a moderator) the U3Og concentrate simply needs to be refined and converted directly to uranium dioxide. Spent reactor fuel is removed, stored, and then either reprocessed or disposed of underground.
Who has tad who mines uranium?
Uranium is widespread in many rocks, and even in seawater. However, like other metals* it is seldom sufficiently concentrated to be economically recoverable. Where it is, we speak of an orebody. In defining what ore is, assumptions are made about the cost of mining and the market price of the metal. Uranium reserves are therefore calculated as tones recoverable up to a certain cost Although it has more than any other country, Australia is not the only one with major deposits. Others in order are: Kazakhstan (15% of world total), Canada, South Africa, Namibia, Brazil, Russia and USA (3%). Many more countries have smaller deposits which could be mined if needed. Uranium is sold only to countries which are signatories of the Nuclear Non-Proliferation Treaty, and which allow international inspection to verify that it is used only for peaceful purposes. Customer countries for Australia's uranium must also have a bilateral safeguards agreement with Australia. Canada has similar arrangements.
Other uses of nuclear energy
Many people, when talking about nuclear energy, have only nuclear reactors (or perhaps nuclear weapons) in mind. Few people realize the extent to which the use of radioisotopes has changed our lives over the last few decades.
Radioisotopes
In our daily life we need food, water and good health. Today, radioactive isotopes play an important part in the technologies that provide us with all three. They are produced by bombarding small amounts of particular elements with neutrons.
In medicine, radioisotopes are widely used for diagnosis and research. Radioactive chemical tracers emit gamma radiation which provides diagnostic information about a person's anatomy and the functioning of specific organs. Radiotherapy also employs radioisotopes in the treatment of some illnesses, such as cancer. More powerful gamma sources are used to sterilize syringes, bandages and other medical equipment. About one in two Australians is likely to experience the benefits of nuclear medicine in their lifetime, and gamma sterilization of equipment is almost universal.
In the preservation of food, radioisotopes are used to inhibit the sprouting of root crops after harvesting, to kill parasites and pests, and to control the ripening of stored fruit and vegetables. Irradiated foodstuffs are accepted by world and national health authorities for human consumption in an increasing number of countries. They include potatoes, onions, dried and fresh fruits, grain and grain products, poultry and some fish. Some prepacked foods can also be irradiated.
In the growing crops and breeding livestock, radioisotopes also play an important rote. They are used to produce high yielding, disease and weather resistant varieties of crops, to study how fertilizers and insecticides work, and to improve the productivity health of domestic animals.
Industrially, and in mining, they are used to examine welds, to detect leaks, to study the rate of wear of metals, and for on-stream analysis of a wide range of minerals and fuels.
There are many other uses. A radioisotope derived from the plutonium formed in nuclear reactors is used in most household smoke detectors.
Radioisotopes are used by police to fight crime, in detecting and analyzing pollutants in the environment, to study the movement of surface water and to measure water runoffs from rain and snow, as well as the flow rates of streams and rivers.
Other reactors
There are also other uses for reactors. Over 200 small nuclear reactors power some 150 ships, mostly submarines, but ranging from icebreakers to aircraft carriers. These can stay at sea for long periods without having to make refueling stops. The wordless first nuclear powered container ship was built in Russia. The heat produced by nuclear reactors can also be used directly rather than for generating electricity. In Sweden and Russia, for example, it is used to heat buildings and to provide heat for a variety of industrial processes such as water desalination.
Military weapons
Both uranium and plutonium were used to make bombs before they became important for making electricity and radioisotopes. But the type of uranium and plutonium for bombs is different from that in a nuclear power plant. Bomb-grade uranium is highly-enriched (>90% U-235, instead of about 3.5%); bomb-grade plutonium is fairly pure (>90%) Pu-239 and is made in special reactors. Today, due to disarmament, a lot of military uranium is becoming available for electricity production. The military uranium is diluted about 25:1 with depleted uranium (mostly U-238) from the enrichment process before being used.
WORDLIST
Abundant богатый
earth's crust земная кора
pitchblende уранинит, смоляная обманка
radioactive decoy радиоактивный распад
rudder (ав.) руль направления
elevator (ав.) руль высоты
shielding защита, защитное покрытие
nuclear fission ядерная реакция деления
chain reaction цепная реакция
nuclear reactor ядерный (атомный) реактор
nuclear power station атомная электростанция (АЭС)
core of a nuclear reactor активная зона ядерного реактора
moderator замедлитель
spent fuel использованное топливо
to be recycled повторно использовать;
рециркулировать
open-cut method открытый способ добычи
bundle сборка (пакет), кассета
disposed fuel отработанное топливо
orebody рудное тело
chemical tracer химический индикатор
prepacked foods расфасованные продукты
on-stream analysis анализ в потоке
smoke detector индикатор(датчик)
aircraft carrier дыма авианосец, авианесущий корабль
refueling пополнение топлива; дозаправка
water desalination опреснение воды
bomb-grade uranium ружейный уран
depleted uranium обедненный уран
EXPRESSIONS:
То grind up (ground up) - размалывать, измельчать в порошок;
another 70 are on the drawing board- другие 70 - в стадии разработки;
to inhibit the sprouting of root crops after harvesting- задерживать прорастание корнеплодов;
to control the ripening of stored fruit and vegetable - регулировать дозревание хранящихся фруктов и овощей;
to examine welds - проверка качества сварных швов;
to study the rate of wear of metals - изучать степень износа металлов.
EXERSISES:
I. Put some questions to the text and translate them:
a) What materials are used for control rods in a reactor core?
b) How did uranium and plutonium get their names?
Please, continue...
II. Translate the following sentences:
a) To prepare uranium for use in a nuclear reactor, it undergoes the steps of mining and milling, conversion, enrichment and fuel fabrication.
b) Uranium is usually mined by either surface (open cut) or underground mining
techniques, depending on the depth at which the ore body is found.
c) The mined uranium ore is sent to a mill which is usually located close to the
mine.
d) U308 is the uranium product which is sold. About 200 tones is required to keep
a large (1000 MW) nuclear reactor generating electricity for one year.
e) The vast majority of all nuclear power reactor in operation and under construction require 'enriched' uranium fuel in which the content of the U-235 isotope has been raised from the natural level of 0.7% to about 3.5%.
f) The first enrichment plants used the gaseous diffusion process, but more modem plants mostly use the centrifuge process. Research is being conducted into laser enrichment, which appears to be a promising new technology.
g) Enriched UF6 is transported to a fuel fabrication plant where it is converted to uranium dioxide (U02) powder and pressed into small pellets.
III. Translate the following material into English:
а) Химический элемент уран: очень твердый тяжелый серебристый радиоактивный металл. В природе обнаруживается только в соединениях, главным образом в смоляной обманке (блестящий минерал, цвет которого меняется от коричневого до чёрного). Играет большую роль в атомной энергетике.
б) Гамма - излучение - магнитное излучение, возникающее при воздействии быстрых заряженных частиц с веществом.
Гамма - лучи подобные рентгеновским, но имеющие более короткую длину волны. Благодаря малой длине волны гамма - лучи обладают очень высокой проникающей способностью, очень малой длинной волны и очень высокой частотой. Они распространяются в воздухе приблизительно на 2,5 км, и являются основной причиной лучевой болезни при использовании атомного оружия.
в) Электрон - это частица, несущая отрицательный заряд. Электроны входят в состав всех атомов и могут также существовать в свободном состоянии.
г) Изотопы являются разновидностью одного и того же химического элемента, которые различаются по массе ядер. Они обладают одинаковыми зарядами ядер (атомным номером), но различаясь числом нейтронов. И имеют одинаковое строение электронных оболочек, т.е. имеют одинаковые химические свойства, и занимают одно и то же место в периодической системе химических элементов.
IV. Translate the following text into Russian:
Uranium Mills and Milling
A uranium mill is a chemical plant designed to extract uranium from mined ore. At conventional mills, the ore arrives via truck and is crushed and leached. In most cases, sulfuric acid is used as the leaching agent, but alkaline leaching can also be done. The leaching agent not only extracts uranium from the ore but also several other constituents; vanadium, selenium, iron, lead, and arsenic. Conventional mills extract 90 to 95 percent of the uranium from the ore. Mills are typically located in areas of low population density, and they process ores (тот mines within about 50 kilometers (30 miles) of the mill.
Atomic EnergyActs defined uranium milling is any activity that results in the production of byproduct material . Byproduct material as it is defined in the Act is "..the tailings or wastes produced by the extraction or concentration of uranium or thorium from any ore processed primarily for its source material content" but adds "...including discrete surface wastes resulting from uranium solution extraction processes."
Potential Hazards:The NRC requirements for uranium mills control industrial hazards and address waste and decommissioning concerns. Because this uranium is not enriched, no criticality hazard, and little fire or explosive hazard, exists for the uranium. The solvent extraction process does present a fire hazard, however. The primary hazards associated with milling operations are occupational hazards found in any metal milling operation that uses chemical extraction plus the chemical toxicity of the uranium itself.
Radiological hazards are low at these facilities as uranium has little penetrating radiation and only moderate non-penetrating radiation. The primary radiological hazard is due to the presence of radium in the uranium decay chains and the production of radon gas from the decay of radium and radon progeny (short-lived radon decay products).
Mill Tailings:The solid (sandy) waste from the conventional uranium milling process is called mill tailings. Uranium mill tailings, which contain most of the progeny of uranium, are a significant source of radon and radon progeny releases to the environment The hazards from radon involve inhalation of radon progeny that may be deposited in the respiratory tract Alpha radiation could be emitted into those tissues and can pose a cancer risk to those workers.
V. Translate the following text into Russian:
Spent fuel storage
Spent fuel assemblies taken from the reactor core are highly radioactive and give off a lot of heat. They are therefore stored in special ponds which are usually located at the reactor site, to allow both their heat and radioactivity to decrease. The water in the ponds serves the dual purpose of acting as a barrier against radiation and dispersing the heat from the spent fuel.
Spent fuel can be stored safely in these ponds for long periods. It can be dry stored in engineered facilities. However, both kinds of storage are intended only as an interim step before the spent fuel is either reprocessed or sent to final disposal. The longer it is stored, the easier it is to handle, due to decay of radioactivity. There are two alternatives for spent fuel:
· Reprocessing to recover the usable portion of it
· Long-term storage and final disposal without reprocessing.
LESSON #3
URANIUM ENRICHMENT
The uranium enriched in uranium-235 is required in commercial light water reactors to produce a controlled nuclear reaction. Several different processes are used to enrich uranium.
Enriching Uranium
Gaseous Diffusion
Gas Centrifuge
Enriching uranium increases the amount of "middle-weight55 and "light-weight" uranium atoms. Not all uranium atoms are the same. When uranium is mined, it consists of heavy-weight atoms (about 99.3% of the mass), middle-weight atoms (0.7%), and light-weight atoms (< 0.01%). These are the different isotopes of uranium, which means that while they all contain 92 protons in the atom's center (which is what makes it uranium). The heavy-weight atoms contain 146 neutrons, the middle-weight contain 143 neutrons, and the light-weight have just 142 neutrons. To refer to these isotopes, scientists add the number of protons and neutrons and put the total after the name: ura-nium-234 or U-234, uranium-235 or U-235, and uranium-238 or U-238.
The fuel for nuclear reactors has to have a higher concentration of U-235 than exists in natural uranium ore. This is because U-235 is the key ingredient that starts a nuclear reaction and keeps it going. Normally, the amount of the U-235 isotope is enriched from 0.7% of the uranium mass to about 5%. Gaseous diffusion is the only process being used in the United States to commercially enrich uranium. Gas centrifuges can also be used to enrich uranium. Although this enrichment process is not used in the United States, the NRC is conducting licensing activities concerning two planned centrifuge facilities.
Gaseous Diffusion
Process: In the gaseous diffusion enrichment plant, the solid uranium hexafluoride (UF6) from the conversion process is heated in its container until it becomes a liquid. The container becomes pressurized as the solid melts and UF6 gas fills the top of the container. The UF6 gas is slowly fed into the plant's pipelines where it is pumped through special filters called barriers or porous membranes. The holes in the barriers are so small that there is barely enough room for the UF6 gas molecules to pass through. The isotope enrichment occurs when the lighter UF6 gas molecules (with the 13-23 and U-235 atoms) tend to diffuse faster through the barriers than the heavier UB6 j molecules containing U-238. One barrier isn't enough, though. It takes many hundreds of barriers, one after the other, before the UF6 gas contains enough uranium-235 used in reactors. At the end of the process, the enriched UF6 gas is withdrawn ft the pipelines and condensed back into a liquid that is poured into containers The UF6 is then allowed to cool and solidify before it is transported to fuel fabrication facilities where it is turned into fuel assemblies for nuclear power reactors.
Hazards: The primary hazards in gaseous diffusion plants include the chemical and radiological hazard of a UF6 release and the potential for mishandling the enriched uranium, which could create a criticality accident (inadvertent nuclear chain reaction).
Gas Centrifuge
The gas centrifuge uranium enrichment process uses a large number of rotating cylinders in series and parallel formations. Centrifuge machines are interconnected to form trains and cascades. In this process, UF6 gas is placed in a cylinder and rotated at a high speed. This rotation creates a strong centrifugal force so that the heavier gas molecules (containing U-238) move toward the outside of the cylinder and the lighter gas molecules (containing U-235) collect closer to the center. The stream that is slightly enriched in U-235 is withdrawn and fed into the next higher stage, while the slightly depleted stream is recycled back into the next lower stage. Significantly more U-23 enrichment can be obtained from a single unit gas centrifuge than from a single unit gaseous diffusion stage. No gas centrifuge commercial production plants are operating in the United States.
Fuel fabrication
Fuel fabrication facilities convert enriched UF6 into fuel for nuclear reactors. Fabrication also can involve mixed oxide (MOX) fuel, which is a combination of uranium and plutonium components. NRC regulates several different types of nuclear fuel fabrication operations.
Types of nuclear fuel fabrications are:
Light Water Reactor Low-Enriched Uranium Fuel
Reactor Mixed Oxide Fuel
Non-Power Reactor Fuel
Other Types of Fuel Fabrication
